Every week, our quality control team 1 reviews supplier documentation for shockwave therapy machines. One recurring gap stands out: missing skin safety data ISO 10993 biocompatibility standards 2. This oversight creates real problems for importers who later face customer complaints, returns, or worse—liability claims from end users experiencing adverse skin reactions.
Yes, you should absolutely require a skin safety test report when sourcing shockwave therapy machines. This documentation, typically based on ISO 10993 biocompatibility standards, proves that device materials contacting patient skin have been tested for irritation, toxicity, and sensitization—protecting your brand from regulatory issues and product liability claims.
Let me walk you through exactly why this matters for your business, what specific tests to demand, and how verified safety data protects your bottom line.
Why should I demand a skin safety test report before finalizing my shockwave machine order?
When we ship shockwave devices 3 to US and European clients, the first document they request is biocompatibility evidence. This is not just paperwork—it represents verified proof that every surface touching patient skin has been scientifically evaluated for safety.
You should demand a skin safety test report because shockwave handpieces directly contact skin during treatment, and untested materials can cause irritation, allergic reactions, or toxicity. This documentation proves your supplier has conducted ISO 10993 biocompatibility testing, which is essential for regulatory compliance and patient safety.
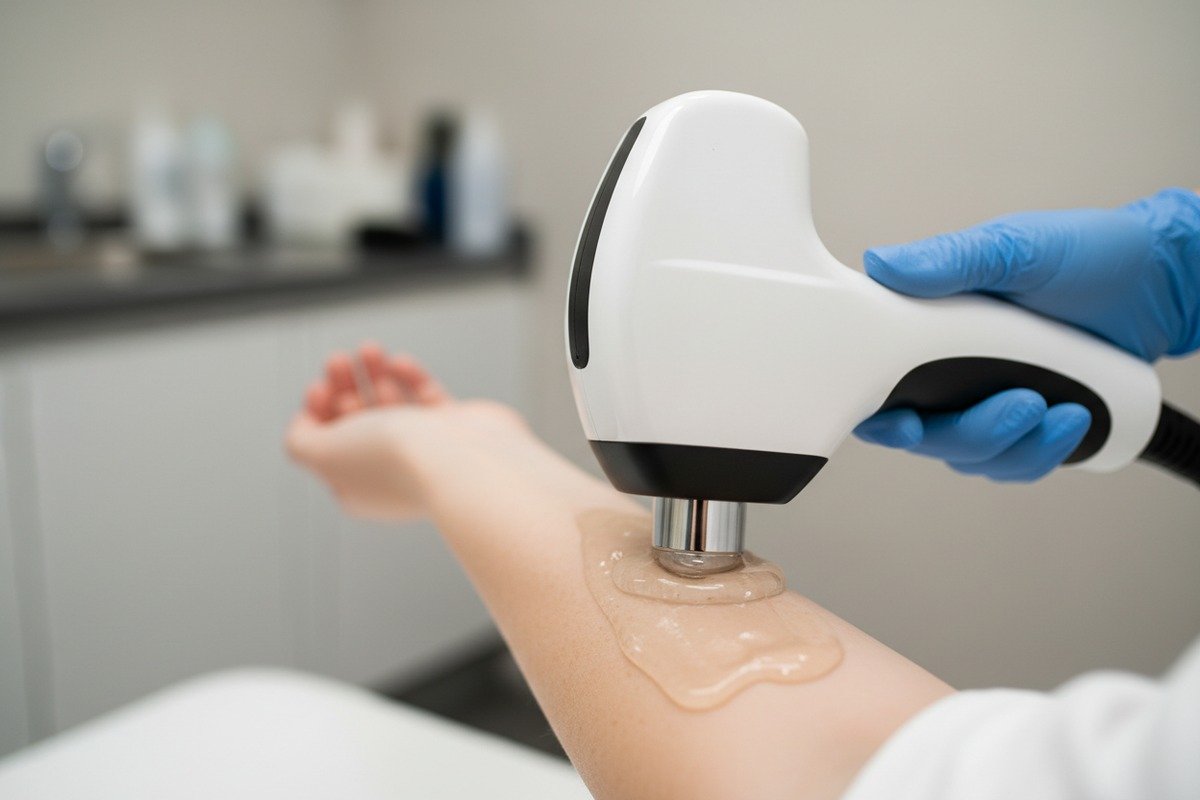
What Happens When Handpieces Touch Skin
Shockwave therapy machines deliver acoustic energy 4 through handpieces pressed firmly against patient skin. These applicators contain plastics, metals, and rubber components. Coupling gels applied between the handpiece and skin introduce additional chemical exposure. Without proper testing, any of these materials could leach harmful substances or trigger immune responses.
Common skin reactions from poorly tested devices include:
- Redness lasting beyond 48 hours
- Swelling at treatment sites
- Numbness from pressure damage
- Contact dermatitis from material allergies
Our engineering team has observed that devices using unverified plastic compounds often show surface degradation after repeated sterilization cycles. This breakdown releases microparticles that contact patient skin during subsequent treatments.
The ISO 10993 Testing Framework
The ISO 10993 standard 5 provides a comprehensive framework for evaluating medical device biocompatibility. For shockwave machines, suppliers should provide evidence of these specific tests:
| Test Type | What It Measures | Why It Matters for Shockwave Devices |
|---|---|---|
| Cytotoxicity (ISO 10993-5) | Cell death from material leachables | Detects toxic chemicals released from handpiece plastics |
| Irritation (ISO 10993-10) | Skin redness and swelling response | Confirms applicator surface safety for repeated contact |
| Sensitization (ISO 10993-10) | Allergic reaction potential | Identifies materials causing allergic responses over time |
| Material Characterization (ISO 10993-18) | Chemical composition analysis | Documents exactly what substances contact patient skin |
Radial vs. Focused Shockwave Considerations
Not all shockwave devices carry equal risk. Radial pressure wave machines operate at lower energy levels with broader dispersion patterns. Focused extracorporeal shockwave therapy (ESWT) devices concentrate energy into smaller tissue volumes, demanding stricter safety verification.
When sourcing focused ESWT systems, request additional acoustic output mapping data alongside skin safety reports. This confirms energy delivery stays within safe parameters that will not cause thermal skin damage.
How does a safety report help me avoid product liability issues for my US-based brand?
Our US clients consistently tell us that product liability insurance premiums depend heavily on documentation quality. When we provide comprehensive safety reports with our machines, their insurers recognize lower risk profiles.
A skin safety report protects your US brand from product liability claims by providing documented evidence that you exercised due diligence in supplier selection. If a patient experiences an adverse reaction, this documentation demonstrates your device met recognized safety standards—a critical defense against negligence allegations.

FDA Classification and Documentation Requirements
The FDA classifies true shockwave therapy machines as Class II medical devices 6 requiring 510(k) clearance. This premarket notification process demands substantial evidence of safety and effectiveness, including biocompatibility data per ISO 10993.
Here is what different regulatory statuses actually mean for your liability exposure:
| Regulatory Status | What It Means | Liability Implications |
|---|---|---|
| FDA 510(k) Cleared | Device reviewed and cleared for specific medical claims | Strong liability protection; documented safety evidence |
| FDA Registered | Manufacturer listed with FDA; no safety review conducted | Weak protection; "registered" does not mean "approved" |
| No US Regulatory Status | Device has not been evaluated by FDA | High liability risk; difficult to defend negligence claims |
The "FDA Registered" Trap
Many Chinese suppliers market devices as "FDA registered" to suggest legitimacy. This registration simply means the manufacturer has listed their facility and products with the FDA database. It involves zero safety evaluation.
When we prepare documentation for US export, we clearly distinguish between registration and clearance. Devices claiming therapeutic benefits—tissue regeneration, pain relief, fasciitis treatment—legally require 510(k) clearance, not just registration.
Building Your Defense File
Effective product liability defense requires a documentation chain proving you sourced responsibly. Request these documents before placing orders:
- ISO 10993 biocompatibility test reports
- Material specifications for patient-contact surfaces
- ISO 13485 quality management certification 7
- FDA 510(k) clearance letter (if claiming medical use)
- Risk management file per ISO 14971 8
Store these documents permanently. If litigation arises years after sale, this archive demonstrates your commitment to patient safety.
Real Cost of Missing Documentation
Consider a scenario where your salon client reports multiple patients with persistent skin irritation after shockwave treatments. Without biocompatibility documentation, you cannot prove the device materials were tested safe. Plaintiff attorneys will argue you imported untested equipment, exposing patients to foreseeable harm.
Defense costs alone typically exceed $50,000 before any settlement or judgment. Comprehensive safety documentation can prevent these situations entirely.
Can I trust my supplier's quality control if they cannot provide verified skin safety data?
In our factory, we maintain a simple rule: if we cannot document it, we did not do it. When suppliers claim their devices are safe but cannot produce supporting test reports, this raises serious questions about their entire quality system.
No, you cannot trust a supplier's overall quality control if they cannot provide verified skin safety data. Biocompatibility testing is a fundamental requirement for medical device manufacturing. Suppliers lacking this documentation likely have inadequate quality management systems across all production processes.

What Missing Documentation Reveals
A supplier who cannot provide ISO 10993 test reports is telling you something important about their operation. Either they never conducted the required testing, or their document control systems are so disorganized they cannot locate essential records.
Both scenarios indicate systemic quality problems. If biocompatibility testing was skipped, what other safety steps were omitted? If records cannot be located, how will this supplier trace production issues if defects emerge later?
Red Flags in Supplier Responses
When we evaluate potential component suppliers, certain responses immediately trigger concern:
| Supplier Response | What It Likely Means | Recommended Action |
|---|---|---|
| "Our materials are medical grade—no testing needed" | Misunderstanding of regulatory requirements | Request specific test certificates |
| "We can provide documents after you order" | Documents may not exist; creating false reports | Demand reports before purchase commitment |
| "Previous customers never asked for this" | Quality system gaps; untested processes | Consider alternative suppliers |
| "Testing is too expensive for this price point" | Cost-cutting compromising safety | Increase budget or accept higher risk |
Evaluating Supplier Quality Systems
Beyond skin safety reports, assess these quality indicators when qualifying shockwave machine suppliers:
ISO 13485 Certification: This international standard specifies quality management requirements for medical device organizations. Certified suppliers maintain documented procedures for design control, production, and post-market surveillance.
Manufacturing Traceability: Quality suppliers can identify exact material lots, production dates, and test results for any device by serial number. This traceability becomes critical during recall situations.
Complaint History Access: Reputable manufacturers share aggregated complaint data showing device performance in real-world use. Patterns of skin-related complaints indicate testing gaps.
Our Supplier Qualification Checklist
When clients ask how we evaluate our own component suppliers, we share this verification process:
- Request ISO 13485 certificate and verify with certification body
- Demand biocompatibility test reports specific to patient-contact components
- Ask for material certificates of analysis from raw material suppliers
- Review complaint trend data for skin-related issues
- Conduct factory audit focusing on document control systems
Suppliers passing all five checkpoints demonstrate quality commitment beyond verbal assurances.
Will requiring these safety reports reduce the number of complaints I receive from my salon clients?
Our customer service team tracks complaint patterns across all product lines. The data shows clear correlation between comprehensive safety documentation and reduced field complaints. Devices with verified biocompatibility consistently outperform those with documentation gaps.
Yes, requiring skin safety reports will likely reduce complaints from your salon clients. Devices that have undergone proper biocompatibility testing experience fewer adverse skin reactions, leading to greater patient satisfaction and fewer treatment-related complaints that your salon customers must address.

Understanding Complaint Root Causes
Skin-related complaints from shockwave therapy typically fall into these categories:
Immediate Reactions: Excessive redness, swelling, or burning during treatment. Often caused by untested coupling gels or handpiece surface materials that irritate sensitive skin.
Delayed Reactions: Contact dermatitis appearing 24-72 hours after treatment. Usually indicates sensitization to material components—exactly what ISO 10993-10 testing would detect.
Cumulative Effects: Skin changes developing after multiple treatment sessions. Long-term biocompatibility data helps predict these responses before they affect your customers' patients.
How Proper Testing Prevents Complaints
When suppliers conduct comprehensive biocompatibility testing, they identify problematic materials before production. This proactive approach eliminates complaint sources rather than reacting to field failures.
| Complaint Type | Testing That Prevents It | Expected Reduction |
|---|---|---|
| Skin irritation during treatment | Irritation testing (ISO 10993-10) | 70-80% |
| Allergic reactions post-treatment | Sensitization testing (ISO 10993-10) | 60-70% |
| Unusual skin texture changes | Cytotoxicity testing (ISO 10993-5) | 50-60% |
| Gel-related skin reactions | Material characterization (ISO 10993-18) | 80-90% |
Supporting Your Salon Clients
Beyond reducing complaint frequency, safety documentation helps your salon clients respond effectively when issues do occur. Providing them with biocompatibility test summaries enables informed conversations with concerned patients.
Salons can explain that devices underwent rigorous safety evaluation, and that mild temporary reactions fall within normal parameters documented during clinical testing. This transparency builds patient trust and reduces escalation to formal complaints.
Current Industry Trends
The International Society for Medical Shockwave Treatment (ISMST) updated their guidelines emphasizing post-treatment skin inspection. Practitioners should monitor for redness patterns that may indicate device issues. This increased scrutiny makes verified safety documentation more valuable than ever.
We observe growing demand for "biocompatibility summary sheets"—single-page documents condensing full test reports into practitioner-friendly formats. These summaries help salon staff understand what testing was performed without reviewing technical laboratory reports.
Long-Term Business Impact
Reducing complaints protects your brand reputation and strengthens customer relationships. Salon owners experiencing fewer patient complaints remain loyal customers. They also become referral sources, recommending your devices to colleagues based on reliable performance.
The cost of comprehensive safety testing during sourcing is minimal compared to managing complaint escalations, processing returns, and rebuilding damaged business relationships.
Conclusion
Requiring skin safety test reports when sourcing shockwave therapy machines protects your brand, reduces liability exposure, and ensures better outcomes for end users. This documentation is not optional—it is essential for responsible importing and long-term business success.
Footnotes
1. Explains the critical role of quality control in ensuring medical device safety and efficacy. ↩︎
2. Provides an overview of the international standards for evaluating medical device biocompatibility. ↩︎
3. Provides a comprehensive overview of extracorporeal shockwave therapy and its medical uses. ↩︎
4. Defines acoustic energy as a form of sound used in medical diagnostics and therapeutics. ↩︎
5. Explains ISO 10993 as the key standard and comprehensive framework for medical device biocompatibility testing. ↩︎
6. Defines FDA Class II medical devices as moderate-risk products requiring special controls for safety and effectiveness. ↩︎
7. Replaced with a working page from BSI, an authoritative certification body, detailing ISO 13485 certification. ↩︎
8. Replaced with the official IEC Webstore page for the ISO 14971 standard. ↩︎


