
I get this question in almost every inquiry from the my customers: do these machines need real clinical validation before hospitals or clinics can use them? Yes, they do.
Yes. Shockwave therapy machines are medical devices. Most markets require clinical validation and safety testing before sale or import. The device must show safe performance, documented risk controls, and credible clinical evidence for the stated indications.
This article explains the common test types, which certifications apply before import, how to verify claims, and what the FDA expects. It uses simple language so teams can brief stakeholders fast.
What clinical tests are commonly required?
I always suggest starting with a test plan that mirrors the approval you want. This saves time and rework down the line.
Common requirements include bench safety and performance tests, biocompatibility for patient-contact parts, electrical safety and EMC, software validation, and clinical evidence for the intended indications. Post-market follow-up is expected after launch.

A complete program combines non-clinical and clinical evidence. Bench tests prove the device hits targets and stays safe. Clinical evidence shows that real patients benefit when trained users follow the instructions.
Core non-clinical tests (typical)
- Electrical safety & EMC: Tests to IEC 60601-1 1 (basic safety) and IEC 60601-1-2 2 (EMC), plus applicable collateral standards.
- Performance & calibration: Verification of energy flux density, frequency accuracy, pulse count accuracy, and stability over duty cycle.
- Acoustic output mapping: Focal depth, beam profile, and repeatability across lots; traceable calibration methods.
- Biocompatibility (ISO 10993): Materials for patient-contact parts tested per ISO 10993 3.
- Usability engineering: Human-factors validation to IEC 62366 4 for screens, workflow, warnings, and lockouts.
- Risk management: Hazard analysis and controls under ISO 14971 5.
Clinical evidence (typical)
- Literature-based clinical evaluation: For well-established indications using equivalent devices with matched technical specs and use conditions.
- Prospective clinical study: When claims are new, parameters differ, or literature is not enough; may be randomized or controlled cohort.
- Real-world PMCF/registry: Post-market data confirming benefit-risk in routine practice.
Table 1 — Typical test types by goal
| Goal | Common test or document | What reviewers look for |
|---|---|---|
| Prove electrical safety | IEC 60601-1 report | Pass results, accredited lab |
| Control emissions/immunity | IEC 60601-1-2 report | Immunity margins, pass/fail logs |
| Verify output | Acoustic/energy bench tests | Tolerance bands, calibration traceability |
| Safe materials | ISO 10993 summary | Contact classification, test matrix |
| Reduce use errors | IEC 62366 file | Validation scenarios, mitigations |
| Show benefit | Clinical evaluation / study | Population, endpoints, stats, AEs |
Clear scope and documented acceptance criteria are vital. Reviewers focus on traceability from risks to tests to results.
Are certifications mandatory before import?
I get asked for a “short list” of required certificates before the freight leaves. That is wise. Missing paperwork at customs is costly.
Yes. Most jurisdictions require specific certifications or registrations before import and sale. Typical items include CE marking under EU MDR for Europe, FDA device listing/clearance for the U.S., and country-specific registrations elsewhere. Safety reports (IEC 60601) and test summaries must support the claim.

For Europe, manufacturers compile Technical Documentation and a Clinical Evaluation Report to obtain CE marking under EU MDR 2017/745 6. In the U.S., most therapeutic models go through the FDA 510(k) program 7 before marketing.
Table 2 — Import readiness snapshot
| Market | Pre-import must-haves | Typical device class |
|---|---|---|
| EU/EEA | CE under MDR + NB certificate | IIa/IIb |
| USA | 510(k) or de novo + listing | II |
| UK | UKCA or accepted CE + MHRA reg | IIa/IIb |
| Canada | MDL + MDSAP | II |
| Australia | ARTG inclusion | IIa/IIb |
If a device ships without these, customs or regulators can hold, fine, or require re-export. Distributors should verify certificates against public databases where available.
How to verify clinical validation reports?
I am often asked, “How can we tell if the clinical file is real and relevant?” A simple, neutral checklist helps both sides.
Verification means checking authenticity, relevance, and traceability. Buyers should confirm accredited test labs, consistent device identity, matched indications, and complete data. Independent literature should support the same parameters.
Confirm the test lab’s accreditation scope to ISO/IEC 17025 8, and validate the Notified Body number in the EU via the NANDO database 9.
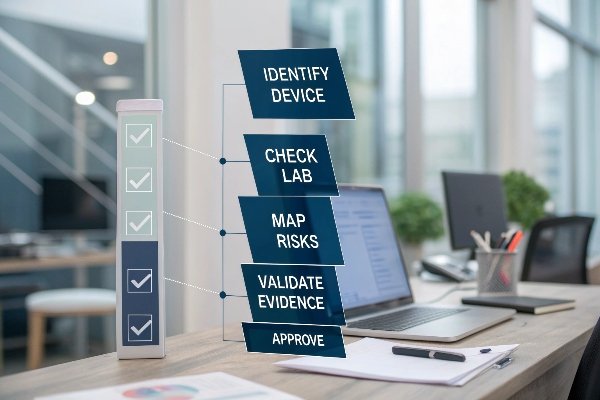
A practical verification workflow
- Identify the device clearly: match trade name, model, hardware version, and software build across all documents.
- Check lab credibility: accreditation and correct standard editions in reports.
- Map risks to tests: align ISO 14971 hazards with verification/validation evidence.
- Validate clinical relevance: confirm equivalence of output ranges, wave type, and patient population.
- Confirm data freshness: current firmware/handpiece generation referenced.
- Cross-check declarations: DoC, NB certificates, or 510(k) summaries list the same model and indications.
Do FDA rules require clinical testing?
I am often asked, “Will the FDA always force us to run new clinical trials?” The answer depends on the device, indication, and predicate.
For many shockwave therapy machines, the FDA pathway is 510(k). Clinical data may or may not be required. If the device matches a predicate in technology and indications, bench and literature evidence can be enough. If claims or technology are novel, clinical studies are often required.

To minimize uncertainty, teams often request a Q-Submission (Pre-Sub) meeting with FDA reviewers to align on evidence expectations 10.
Table 3 — When the FDA often expects clinical data
| Situation | Likelihood of clinical data need | Notes |
|---|---|---|
| Same tech + same indication as predicate | Low | Strong bench + literature may suffice |
| New indication, same tech | Medium | Targeted clinical evidence likely |
| New tech features (e.g., combined modality) | High | Clinical + extended bench work |
| Expanded superiority claims | High | Prospective comparative data |
Conclusion
Yes, clinical validation matters. Plan bench tests, safety standards, and the right evidence for the chosen market. Verify reports carefully, align claims with approvals, and use early FDA or Notified Body feedback to stay on track.
Footnotes
1. IEC 60601-1 — Core electrical/mechanical safety standard for medical devices. ↩︎
2. IEC 60601-1-2 — Electromagnetic compatibility requirements and tests. ↩︎
3. ISO 10993 — FDA-recognized framework for biocompatibility evaluation. ↩︎
4. IEC 62366 — Usability engineering to reduce use-related risks. ↩︎
5. ISO 14971 — Risk management process for medical devices. ↩︎
6. EU MDR 2017/745 — Legal basis for CE marking and clinical evaluation. ↩︎
7. FDA 510(k) — Database to verify substantial equivalence clearances. ↩︎
8. ISO/IEC 17025 — Accreditation for test/calibration laboratory competence. ↩︎
9. NANDO — EU directory to verify Notified Bodies and scopes. ↩︎
10. FDA Q-Submission — Process to get premarket feedback on evidence plans. ↩︎


