Every week, our production team ships dozens of shockwave therapy machines 1 to clinics worldwide. Yet the question we hear most often surprises us: “Can I actually use this without a doctor on staff?” The confusion is real. Regulations vary wildly. Risks exist. And wrong answers could mean legal trouble or patient harm.
Whether you need a doctor or therapist depends on your location, device type, and intended use. Professional-grade shockwave machines typically require licensed healthcare oversight in most regions, while low-energy home devices face fewer restrictions. Always verify local regulations before offering treatments.
This guide breaks down the legal requirements, certification needs, training protocols, and supervision options for operating shockwave therapy equipment CE marking authorities 2. Let us walk you through everything you need to know before making your decision.
Can I legally use a shockwave therapy machine in my clinic without a medical license?
Running a clinic means understanding where the legal lines are drawn. Our export team has seen clients face unexpected hurdles because they assumed regulations were universal. They are not. What works in Germany may violate laws in California.
In most jurisdictions, operating professional shockwave therapy machines in a clinical setting requires some form of medical licensure or healthcare credential. The specific requirements depend on your country, state, and the classification of your device. Unlicensed operation can result in fines, lawsuits, or clinic closure.

Understanding Device Classification
The legal landscape starts with how your device is classified. Regulatory bodies like the FDA in the United States 3 and CE marking authorities in Europe categorize medical devices by risk level.
Professional shockwave therapy machines fall under stricter categories. In the US, the FDA typically classifies them as Class II or Class III medical devices. These require clinical oversight. Home wellness devices often dodge these classifications by marketing as "non-medical" products with lower energy outputs.
| Device Type | Typical Classification | Oversight Required | Energy Output |
|---|---|---|---|
| Professional Clinical | Class II/III (FDA) or Medical CE | Licensed practitioner | High (calibrated mJ/mm²) |
| Home Wellness | Consumer product | None mandated | Low (uncalibrated) |
| Hybrid/Portable Pro | Varies by region | Often required | Medium to high |
Regional Variations in Requirements
Our clients in Europe face Medical Device Regulation 4 requirements that demand professional oversight for clinical devices. The General Safety and Performance Requirements specify who can operate such equipment.
In the United States, state medical boards govern practice scope. A chiropractor in Texas may legally offer shockwave therapy, while an aesthetician in New York may not. Physical therapists, podiatrists, and physicians generally have clearer pathways to legal use.
Canada follows similar patterns with provincial licensing boards. Australia requires devices to be on the Australian Register of Therapeutic Goods 5 for clinical use.
The Consequences of Non-Compliance
Operating without proper credentials carries real risks. We have heard from prospective clients who faced cease-and-desist orders after competitors reported them. Others dealt with malpractice claims 6 when treatments went wrong without licensed supervision.
Insurance companies may deny coverage for incidents involving unlicensed operators. Professional liability policies often specify credential requirements in their terms.
The smart approach is consulting a healthcare attorney in your jurisdiction before purchasing equipment. The cost of legal advice is far less than the cost of shutting down your business.
Do my employees need a specific certification to operate this device on my clients?
When our training specialists work with new buyers, employee qualifications always come up. Clinic owners want to maximize their investment by having multiple staff members operate the equipment. But can your receptionist deliver shockwave treatments? The short answer: probably not.
Most jurisdictions require device operators to hold relevant healthcare credentials or work under direct supervision of licensed practitioners. Specific certification requirements vary, but unlicensed staff typically cannot independently administer shockwave therapy. Check your local scope-of-practice laws before delegating treatments.
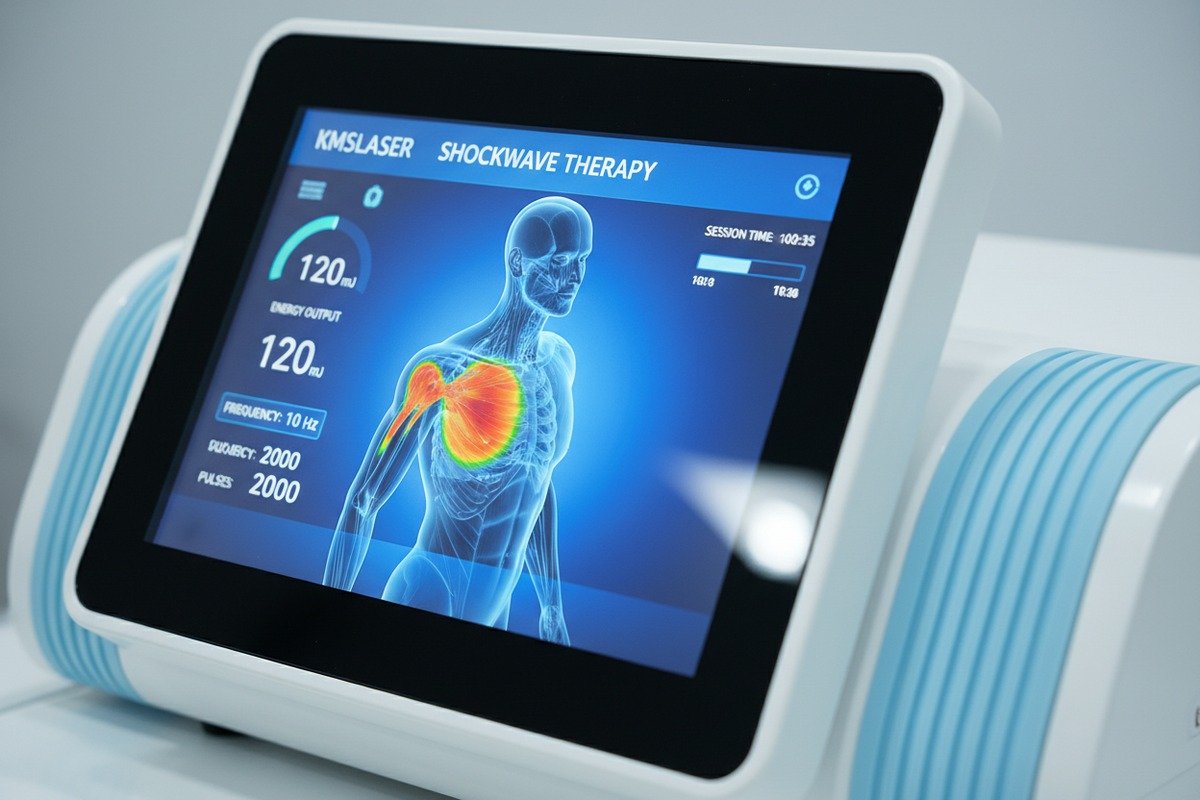
Who Can Legally Operate the Equipment?
The operator requirements depend on two factors: your location and the treatment type. Generally, these professionals can use clinical shockwave devices:
- Licensed physicians and surgeons
- Physical therapists and physiotherapists
- Chiropractors
- Podiatrists
- Sports medicine specialists
- Licensed massage therapists (in some regions, with limitations)
Our engineering team designs devices with these users in mind. The interfaces assume baseline medical knowledge. Understanding anatomy, contraindications 7, and treatment protocols matters for safe operation.
Certification Programs and Training
Many device manufacturers offer certification programs. When we ship machines, our team includes training materials and can arrange video instruction sessions. However, manufacturer certification differs from legal licensure.
| Credential Type | What It Covers | Legal Standing |
|---|---|---|
| Manufacturer Certification | Device operation, protocols | Often required by warranty, not legally mandated |
| Professional License | Scope of practice, medical training | Legally required for clinical practice |
| Continuing Education | Specialized technique courses | May satisfy license renewal requirements |
| State-Specific Certification | Regional requirements | Mandatory in some jurisdictions |
Some states in the US now offer specific shockwave therapy certifications. Florida and California have continuing education requirements that cover this modality. Physical therapy boards often include it under their scope without separate certification.
Delegation and Supervision Models
If your licensed staff cannot personally deliver every treatment, supervision models exist. A physician can often delegate to trained assistants who work under their license. The supervising practitioner remains liable for outcomes.
Direct supervision typically means the licensed professional is physically present. General supervision may allow them to be available but not in the room. Indirect supervision involves review of treatment plans without presence during procedures.
Our recommendation: document everything. Written protocols, supervision agreements, and training records protect everyone if questions arise later.
How can I ensure my staff is properly trained to avoid safety risks and misuse?
Our quality control team catches defects before shipping. But even a perfect machine causes harm in untrained hands. We have seen burns, bruises, and worse when operators skip steps or ignore contraindications. Proper training is not optional. It is your first line of defense against liability.
Comprehensive staff training should cover device mechanics, treatment protocols, contraindication screening, emergency procedures, and ongoing competency assessments. Partner with your device manufacturer for initial training, supplement with professional courses, and implement regular skills verification. Document all training thoroughly.
Essential Training Components
When our technical specialists train new users, they follow a structured curriculum. Your internal training should address these areas:
Anatomical knowledge comes first. Operators must identify treatment targets accurately. Missing the mark reduces efficacy and risks damaging healthy tissue. Understanding musculoskeletal anatomy at a functional level is mandatory.
Device mechanics matter too. Our machines use touchscreen interfaces with anatomical diagrams and technical readouts. Staff need to interpret these displays correctly. They must adjust energy levels, pulse frequencies, and applicator pressure appropriately.
Contraindication Screening Protocols
This area causes the most problems. Operators must screen every client before every session. Contraindications include:
- Pregnancy
- Blood clotting disorders or anticoagulant use
- Active infections in treatment area
- Malignant tumors
- Recent fractures
- Implanted devices (pacemakers, defibrillators)
- Open wounds
- Nerve disorders affecting treatment site
| Contraindication Category | Examples | Action Required |
|---|---|---|
| Absolute | Pregnancy, cancer at site, pacemaker | Refuse treatment entirely |
| Relative | Anticoagulants, diabetes, elderly patients | Physician consultation first |
| Temporary | Active infection, recent injury | Delay until resolved |
Create intake forms that capture this information. Update them regularly. Train staff to ask follow-up questions when answers seem incomplete.
Practical Skills Development
Theory without practice leads to mistakes. Our recommendation is a graduated training approach. New operators should observe experienced staff first. Then they practice on themselves or colleagues before treating clients. Finally, supervised client sessions build confidence before independent work.
Energy progression matters for safety. Starting at low settings and gradually increasing prevents tissue damage. Sessions typically run 15-20 minutes with gel application for transmission. Staff must learn appropriate pressure and movement patterns for different body areas.
Ongoing Competency Verification
Initial training fades without reinforcement. Implement quarterly skills assessments. Review treatment logs for anomalies. Encourage staff to report near-misses without punishment. These create a culture of continuous improvement.
When we update our machines with new features, we provide refresher training materials. Your staff should review these promptly. Technology evolves, and yesterday's best practices may become obsolete.
Can I use a licensed therapist instead of a doctor to supervise my shockwave treatments?
Our clients often ask whether they need a full physician on staff or if other licensed professionals suffice. The cost difference is substantial. Doctors command higher salaries. Therapists are more accessible. Understanding your options saves money without sacrificing compliance.
Yes, in most jurisdictions, licensed therapists such as physical therapists, chiropractors, and podiatrists can legally supervise and administer shockwave therapy without physician oversight. The key is that the supervising professional's license must include shockwave therapy within its scope of practice. Verify with your local licensing board.

Scope of Practice by Profession
Different healthcare credentials carry different treatment authorities. Understanding these distinctions helps you structure your clinic appropriately.
Physical therapists and physiotherapists have the broadest access in most regions. Shockwave therapy falls naturally within their musculoskeletal treatment mandate. They can typically operate independently without physician supervision.
Chiropractors also generally qualify. Their scope covers neuromusculoskeletal conditions where shockwave therapy applies. State-by-state variations exist, so verify locally.
Podiatrists treat foot and ankle conditions. Plantar fasciitis 8 and Achilles tendinopathy are prime shockwave therapy applications. Most podiatric licenses cover this modality.
| Professional | Typical Shockwave Authority | Common Limitations |
|---|---|---|
| Physical Therapist | Full independent authority | May need referral for diagnosis |
| Chiropractor | Full independent authority | Varies by state/province |
| Podiatrist | Lower extremity focus | Treatment area restrictions |
| Athletic Trainer | Often requires supervision | Cannot diagnose |
| Massage Therapist | Usually prohibited | Scope too limited |
The Diagnosis Question
Here is where complications arise. Shockwave therapy works best after proper diagnosis. Many therapists can treat conditions but cannot diagnose them independently.
A physical therapist may legally operate the machine. But determining that a patient has tendinopathy rather than a stress fracture may require physician assessment first. This creates practical workflows where doctors diagnose and therapists treat.
Our export clients often establish referral networks. A physician evaluates new patients, confirms diagnosis, and refers appropriate cases to therapists for ongoing treatment. This model satisfies legal requirements while containing costs.
Insurance and Reimbursement Considerations
Legal authority to treat does not guarantee payment. Insurance reimbursement policies vary by provider credential. Some insurers only cover shockwave therapy when administered by physicians. Others accept qualified therapists.
Before building your service around therapist-delivered care, verify that your target insurers will pay. Cash-pay models avoid this complexity but limit your market.
Building a Compliant Supervision Structure
If you want non-licensed staff involved in any capacity, establish clear supervision protocols. Document the chain of responsibility. The supervising therapist or physician must review treatment plans, remain accessible during procedures, and sign off on patient records.
Our quality management system emphasizes documentation. Yours should too. Written protocols protect you when regulators or attorneys come asking questions. "We followed our documented procedures" is a strong defense.
Conclusion
Navigating shockwave therapy regulations requires attention to your specific situation. Professional devices need qualified operators. Training prevents harm. Therapists often suffice instead of physicians. When in doubt, consult local licensing boards and healthcare attorneys before investing. Your compliance protects both your patients and your business.
Footnotes
1. Authoritative overview of extracorporeal shockwave therapy. ↩︎
2. Explains the definition, requirements, and process for CE marking of medical devices in the EU. ↩︎
3. Official FDA overview of medical device regulation. ↩︎
4. Official European Medicines Agency page on medical device legislation and regulations. ↩︎
5. Official Australian government database for therapeutic goods, including medical devices. ↩︎
6. Provides a general definition and overview of medical malpractice as a legal cause of action. ↩︎
7. Authoritative medical encyclopedia definition of contraindications from a US government site. ↩︎
8. Authoritative medical information on symptoms, causes, and treatment of plantar fasciitis. ↩︎


