
From working closely with clinics and distributors, we see the same question again and again: “Can anyone operate this machine, or do I need a professional licence to use it on patients?”
Operating a shockwave therapy machine usually requires a licensed healthcare professional, or at least falls under the scope of a licensed practitioner’s responsibility, but exact licensing rules depend on the country, professional board regulations, and whether the device is marketed for medical or purely aesthetic indications.
For buyers, the safest strategy is to assume that shockwave therapy is a regulated clinical service 1, not a casual spa add-on, and then confirm specific rules with local authorities.
What qualifications are typically needed to use shockwave machines?
From our perspective as a manufacturer supplying both hospitals and private clinics, most of our serious buyers already hold a healthcare credential and simply want to know whether their qualification is sufficient for shockwave therapy.
In most healthcare systems, shockwave therapy is performed by licensed professionals such as physiotherapists 2, chiropractors, physicians, podiatrists, or other regulated clinicians, often after completing additional device-specific training or continuing-education courses 3.
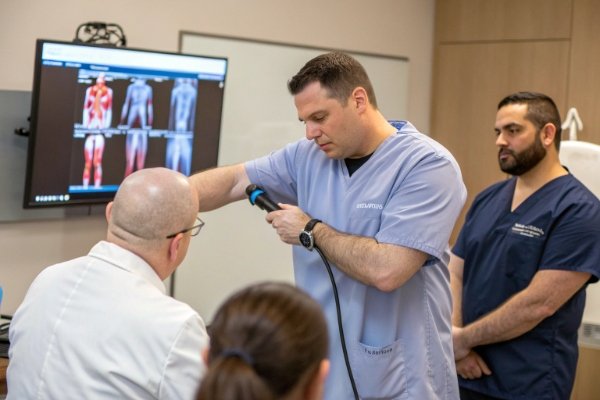
The exact job title varies by country, but the pattern is similar: regulators expect the operator to have a recognised clinical background plus training in shockwave use.
Typical professional backgrounds
In medical and rehabilitation settings, shockwave therapy is commonly delivered by:
- Physiotherapists / physical therapists 4
- Sports medicine physicians
- Orthopaedic or rehabilitation doctors
- Chiropractors 5
- Podiatrists
- Osteopaths
- Sometimes advanced practice nurses or physician assistants (depending on local rules)
Training beyond a basic licence
Holding a licence does not automatically mean someone understands shockwave parameters, indications, or contraindications. That is why many clinics require:
- Manufacturer training on the specific model
- Formal CPD/CEU courses in shockwave therapy
- Internal competency sign-offs before independent use
Typical qualification matrix
| Role / Background | Can Use Shockwave?* | Notes |
|---|---|---|
| Medical doctor (orthopaedics, sports, rehab) | Yes, usually | Often prescribes and may personally apply |
| Physiotherapist / physical therapist | Yes, very common | Often main operator in rehab settings |
| Chiropractor | Yes, where scope permits | Common in musculoskeletal practices |
| Podiatrist | Yes, for foot and ankle conditions | Widely used for plantar fasciitis 6 |
| Massage therapist (no medical licence) | Depends on local rules | Often not allowed without clinical licence |
| Beauty therapist / aesthetician | Only for non-medical indications, where allowed | Must follow cosmetic regulations and local law |
*Always subject to each country’s professional regulations and scope of practice.
Clinics that treat shockwave like a purely commercial “machine service” without verifying qualifications put themselves at regulatory and legal risk. It is safer to start from a licensed, clinically trained operator and build protocols from there.
How do licensing requirements vary across regions and use cases?
Because we ship devices to many countries, we see how dramatically rules can change as soon as you cross a border—or change your marketing message from “medical treatment” to “aesthetic body contouring.”
Licensing requirements vary by region based on national healthcare laws, scope-of-practice definitions, and whether the shockwave device is used for medical diagnoses, therapeutic indications, or purely aesthetic purposes 7, so buyers must always check local rules rather than copying another country’s model.

Regional patterns (general, not legal advice)
-
North America
Shockwave is typically considered a medical or therapeutic modality. Operators usually must be licensed healthcare providers (MD, PT, DC, etc.), working within their scope of practice. -
European Union / UK
MDR regulates the device 8, while national professional laws regulate the person who may use it. Many countries limit use to physicians, physiotherapists, or other recognised healthcare professionals. -
Asia-Pacific & Middle East
Rules vary widely. Some regions follow EU-style patterns; others have looser enforcement, especially in wellness and aesthetic settings.
Medical vs aesthetic positioning
Licensing expectations often change depending on how the device is positioned:
-
Medical indication (e.g., plantar fasciitis, tendinopathy):
Usually reserved for licensed clinicians with diagnostic authority or close collaboration with such clinicians. -
Aesthetic indication (e.g., cellulite, body contouring, local circulation improvement):
Sometimes allowed in beauty clinics under less strict professional-licence rules, but still subject to consumer safety and advertising laws.
Region and indication overview
| Region / Market | Medical Use (e.g., tendinopathy) | Aesthetic Use (e.g., cellulite) |
|---|---|---|
| US / Canada | Licensed health professionals only | Varies; many states still expect supervision |
| EU (general pattern) | Regulated clinicians, per national law | Some uses allowed in aesthetics, country-specific |
| UK | Health professionals via HCPC / GMC | Beauty sector under general consumer law |
| Middle East | Often doctor-led clinics | Aesthetic use sometimes in beauty centres |
| Asia-Pacific | Mixed; country-specific licensing | Often more flexible, but changing over time |
Because interpretations change and regulators update guidance, your safest approach is to treat shockwave therapy as a clinical-grade technology and then confirm local specifics with your professional board or health authority.
Can non-licensed staff use these machines under supervision?
Many of our customers run busy centres with support staff, so they often ask whether assistants or non-licensed personnel can operate the shockwave machine while a licensed clinician oversees the case.
In many jurisdictions, non-licensed staff may assist with shockwave therapy 9 under the direct supervision of a licensed professional, but ultimate responsibility remains with the supervising clinician, who must ensure appropriate assessment, parameter selection, and safety monitoring.

The legal details differ, but a few consistent principles appear across markets.
Typical division of responsibilities
-
Licensed clinician
– Performs assessment and diagnosis
– Decides if shockwave is indicated
– Selects treatment area and initial settings
– Monitors response and adjusts protocol
– Accepts clinical and legal responsibility -
Assistant or therapy aide
– Prepares the room and patient
– Applies gel, positions patient
– May operate the handpiece within set parameters, if permitted locally
– Reports any discomfort or unusual reactions immediately
Levels of supervision
Different systems define supervision levels, such as:
- Direct supervision – clinician is in the room or immediate area
- On-site supervision – clinician present in the facility and available
- Remote / general supervision – often not acceptable for first-time or high-energy treatments
Supervision framework table
| Role | Allowed Activity (typical) | Supervision Level Expected |
|---|---|---|
| Licensed clinician | Full assessment and treatment | Independent practice |
| Licensed clinician + aide | Clinician sets plan; aide may assist application | Direct or on-site supervision |
| Non-licensed alone | Usually not allowed for therapeutic indications | High legal and ethical risk |
Even where the law does not explicitly forbid an assistant from holding the handpiece, insurers and professional associations often expect a licensed clinician to maintain close oversight. From a risk-management standpoint, leaving shockwave treatments entirely to non-licensed staff is rarely a wise choice.
What documentation should suppliers provide for regulatory guidance?
Because we work with buyers entering new markets, we are often asked to supply not just a machine, but also orientation on how to use it legally and safely under different regulatory frameworks.
Suppliers should provide clear documentation including regulatory status (e.g., CE, FDA), indications for use, contraindications, recommended operator profile, and written guidance stating that users must follow local licensing and scope-of-practice rules when delivering shockwave therapy.

While suppliers cannot replace a local lawyer or licensing board, they can make it much easier for you to understand where the device sits in the regulatory landscape.
Key documents to request from suppliers
-
Regulatory clearance summary
– CE certificates 10 or EU MDR declaration
– FDA 510(k) or equivalent (where applicable)
– Other national approvals (e.g., Health Canada, TGA, etc.) -
Indications for use document
– Exactly which conditions the device is cleared for
– Whether the approvals are medical or aesthetic -
Operator suitability statement
– Recommended minimum background (e.g., licensed healthcare professional)
– Training requirements
– Restrictions for home or non-professional use -
Clinical and safety manual
– Contraindications, precautions, and side effects
– Dosage guidelines and parameter ranges
– Advice on patient selection and follow-up
Documentation overview table
| Document Type | Why It Matters | What You Should Check |
|---|---|---|
| Regulatory approvals | Shows legal status of the device | Class, market, and intended use |
| Indications for use | Defines medical vs aesthetic positioning | Fit with your scope of practice |
| Operator profile guidance | Clarifies who should use the device | Aligns with your team’s qualifications |
| Safety / clinical manual | Supports protocols and staff training | Clear contraindications and warnings |
A supplier who understands compliance will proactively share this information and emphasize that the final responsibility for legal operation lies with the local clinic and its licensed professionals.
Conclusion
Operation of a shockwave therapy machine 11 is usually limited to licensed healthcare professionals, with any involvement of non-licensed staff requiring close supervision and clear protocols, supported by supplier documentation and careful checking of local licensing and scope-of-practice rules.
Footnotes
1. Understand the necessary protocols and competency required to integrate shockwave therapy into clinical practice. ↩︎
2. Overview of how extracorporeal shockwave therapy (ESWT) is applied within physiotherapy practice worldwide. ↩︎
3. View examples of Continuing Professional Development (CPD) courses available for shockwave therapy training. ↩︎
4. Reference to a US Physical Therapy Board ruling that affirms shockwave therapy is within the scope of practice for physical therapists. ↩︎
5. California state regulations detailing the authorized scope of practice for licensed chiropractors. ↩︎
6. Detailed clinical overview of plantar fasciitis and how Extracorporeal Shock Wave Therapy (ESWT) is used to treat it. ↩︎
7. Comparative study on how different global regulatory bodies classify and regulate aesthetic devices versus medical devices. ↩︎
8. Comprehensive explanation of the European Union Medical Device Regulation (MDR) and its mandatory compliance requirements. ↩︎
9. Official US Federal Register entry on the classification of Extracorporeal Shock Wave devices for plastic surgery (aesthetic) use. ↩︎
10. European Medicines Agency page detailing the regulatory framework for medical devices in the EU. ↩︎
11. Insights from a medical institution on the applications and ongoing research into Extracorporeal Shockwave Therapy (ESWT) in musculoskeletal medicine. ↩︎


