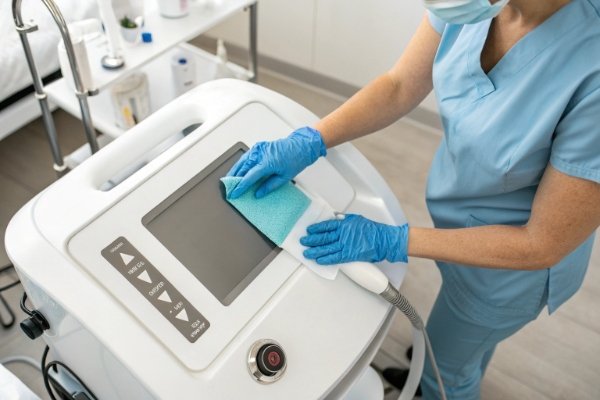
From our experience supplying machines to clinics, spas, and physiotherapy centers, one of the most common practical questions is how simple it is to maintain daily hygiene between clients.
Most shockwave therapy machines are designed to be easy to clean, using smooth surfaces, detachable transmitters, and wipe-friendly components—but the real level of convenience depends on the machine’s structure, the materials used in patient-contact parts, and the cleaning rules recommended by the supplier 1.
A clear understanding of cleaning workflow helps clinics manage hygiene standards without slowing treatment schedules.
What design elements simplify machine cleaning and hygiene?
Whenever we work with clinics to set up their devices, they often focus on how quickly staff can turn the room over between appointments. This depends heavily on design.
Good machine design makes cleaning fast by including smooth housings, detachable transmitters, sealed control panels, rounded edges, gel-resistant surfaces, and handpieces built for quick disassembly without tools—reducing downtime and lowering the risk of contamination 2.
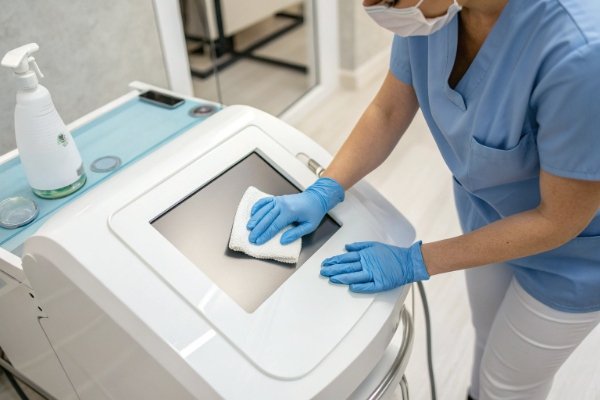
Below is a breakdown of design choices that make cleaning easier for staff working under time pressure.
Smooth and sealed device body
Smooth plastic or metal enclosures allow wipe-down in seconds.
Sealed buttons or touchscreens prevent fluid from entering the control panel.
Flat surfaces reduce gel accumulation.
Detachable transmitters
Most radial devices include screw-on or quick-release transmitters to clean after each client.
Focused units often include removable focusing heads for deeper cleaning.
Gel-resistant construction
Shockwave treatments use gel 3, so well-designed devices are built to prevent:
- gel seepage into seams
- buildup around buttons
- sticky residue on handpiece joints
Easy-access handpiece architecture
Clinics benefit from handpieces that allow quick partial disassembly:
- transmitter removal
- O-ring access
- projectile cleaning (radial)
- barrel wiping
Table — Design elements that improve hygiene workflow
| Design Feature | Cleaning Advantage | Seen On |
|---|---|---|
| Smooth chassis | Fast wipe-down without residue | Most mid/high-end devices |
| Sealed touchscreen | Prevents fluid entry; simple sanitation | Premium models |
| Detachable transmitters | Enables patient-by-patient cleaning | Radial & focused |
| Quick-open handpiece | Faster weekly cleaning | High-quality radial |
| Rounded edges | No fluid traps | Medical-grade devices |
Why these features matter
Short sentences improve clarity.
Clinics save time between appointments.
Staff follow hygiene guidelines more easily.
The device stays in better long-term condition.
Patient trust increases when cleaning looks professional.
Are detachable parts or covers provided for sanitation?
One of the most common questions we receive from clinics is whether patient-contact components can be removed for deep cleaning.
Most shockwave therapy machines include detachable transmitters, removable O-rings, and serviceable handpiece components, and some units even offer protective covers to reduce daily cleaning time—though the number of detachable parts varies by brand and device class 4.

Below is an overview of what clinics can usually expect.
Common detachable parts
- Transmitters (radial): unscrew for cleaning
- Applicator heads (focused): wipe or remove depending on model
- O-rings: clean gel residue regularly
- Handpiece barrels: open weekly for dust and debris removal
- Projectiles (radial): removable for periodic cleaning
- Silicone sleeves/caps: used in cosmetic models to reduce gel mess
Optional protective accessories
Some manufacturers supply:
- disposable covers
- silicone protective rings
- washable handpiece sleeves
- gel guards
These reduce cleaning time between clients.
Table — Typical detachable sanitation components
| Component | Purpose | Cleaning Frequency |
|---|---|---|
| Transmitters | Main patient-contact part | After each client |
| O-rings | Seal for gel and impact protection | Daily |
| Barrel interior | Shockwave projectile chamber | Weekly |
| Projectiles (radial) | Improves impact efficiency | Weekly/monthly |
| Silicone caps/sleeves | Keeps handpiece exterior clean | Replace or sanitize |
| Cable connectors | Occasionally cleaned externally | Weekly |
Why detachable parts matter
Quick removal allows fast sanitation.
Broken-down parts dry faster after cleaning.
Technicians avoid gel buildup on internal mechanisms.
Hygiene protocol becomes more predictable.
What cleaning agents are recommended or restricted by suppliers?
Clinics often ask what they can safely use, because harsh chemicals can damage the machine or void the warranty.
Most suppliers recommend mild soap, alcohol wipes 5, medical-surface disinfectants, or gel removers—while restricting corrosive chemicals such as bleach, acetone, ammonia, and industrial cleaners that can degrade handpiece materials, O-rings, and plastic housings.

Below are details on what is commonly allowed or prohibited.
Recommended cleaning agents
- Mild antibacterial soap
- Diluted neutral detergent
- 70% isopropyl alcohol wipes
- Ultrasound gel-removal spray
- Medical-grade surface disinfectants
These are gentle on plastics, aluminum housings, and silicone parts.
Restricted or prohibited agents
- Bleach 6
- Acetone
- Ammonia-based cleaners
- Industrial degreasers
- Abrasive powders
- High-concentration alcohol (>90%)
These can degrade coatings and cause drying or cracking of seals.
Table — Cleaning agent compatibility
| Cleaning Agent | Allowed? | Notes |
|---|---|---|
| Mild soap solution | Yes | Safe for daily use |
| 70% isopropyl alcohol | Yes | Most common disinfectant |
| Medical surface spray | Yes | Check brand-specific rules |
| Bleach | No | Corrodes metal and plastics |
| Acetone | No | Damages housing and O-rings |
| Ammonia cleaners | No | Breaks down surface coatings |
Why the right agents matter
Correct cleaning products protect the handpiece.
They keep O-rings flexible.
They extend the machine’s life.
They prevent discoloration or cracking.
They safeguard warranty protection.
How often should users clean different machine components?
Because we train many clinics during onboarding, we see how important a structured cleaning schedule is to daily workflow.
Most clinics clean transmitters after each client, wipe the machine body daily, open the handpiece weekly, and perform deep cleaning after a set number of pulses—often every 20,000 shots or as recommended by the supplier’s protocol 7.
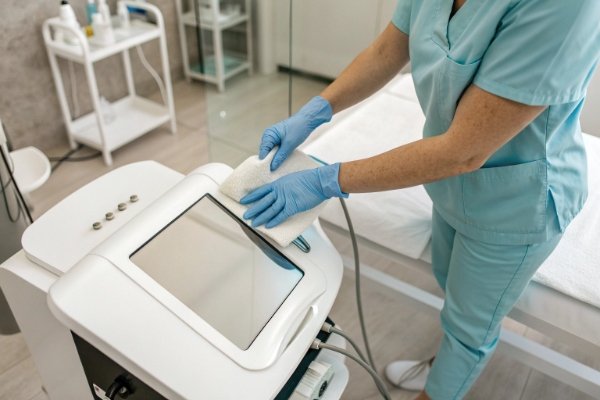
Below is a clear schedule that clinics commonly follow.
Cleaning after each client
- Remove gel from transmitters
- Wipe handpiece exterior
- Clean O-rings
- Sanitize patient-contact surfaces
Takes about 2–5 minutes.
Daily cleaning
- Wipe device body
- Clean touchscreen or button panel
- Inspect cables
- Remove gel from connectors
Weekly cleaning
- Open radial handpiece barrel
- Clean projectile and chamber
- Check springs and O-rings for wear 8
- Clean interior dust buildup
Long-interval maintenance
Many manufacturers recommend deep cleaning:
- Every 20,000 pulses (whichever comes first)
- Or monthly, depending on clinic volume
Table — Cleaning schedule overview
| Component | Frequency | Notes |
|---|---|---|
| Transmitters | After each use | Fast cleaning essential |
| Handpiece exterior | Daily | Keeps gel from hardening |
| Barrel interior | Weekly | Prevents dust and debris |
| Projectiles (radial) | Weekly/monthly | Improves impact efficiency |
| Device body | Daily | General hygiene |
| Deep cleaning cycle | Pulse-based | Follows manufacturer rules |
Why routine cleaning matters
It protects internal components.
It prevents gel hardening.
It reduces handpiece replacement costs.
It helps maintain consistent energy output 9.
It shows professionalism to your clients.
Conclusion
Shockwave therapy machines are built for quick, efficient cleaning. Smooth surfaces, detachable transmitters, easy-access handpieces, and approved cleaning chemicals all help clinics maintain high hygiene standards 10 with minimal downtime.
Footnotes
1. General guidelines for infection control and cleaning procedures recommended by major medical device suppliers. ↩︎
2. CDC guidelines on cleaning protocols necessary to lower the risk of contamination in healthcare settings. ↩︎
3. Research detailing the chemical composition of ultrasound gels and the importance of removing residue post-treatment. ↩︎
4. FDA’s regulatory information on the classification of medical devices and how it affects manufacturing standards and material selection. ↩︎
5. Study on the effectiveness and safety of alcohol wipes for surface disinfection, specifically in clinical environments. ↩︎
6. CDC guidelines on disinfectants, clarifying which chemicals, like bleach, should be restricted due to corrosive properties. ↩︎
7. Overview of maintenance protocols and schedules necessary to ensure the long-term operational efficiency of industrial and medical equipment. ↩︎
8. Technical recommendations from the International Society for Medical Shockwave Treatment regarding the routine maintenance of handpiece components. ↩︎
9. Research emphasizing the importance of maintenance in ensuring that energy delivery from therapeutic devices remains consistent across uses. ↩︎
10. Detailed CDC recommendations for cleaning and reprocessing non-critical patient-care medical devices to meet clinical hygiene standards. ↩︎


