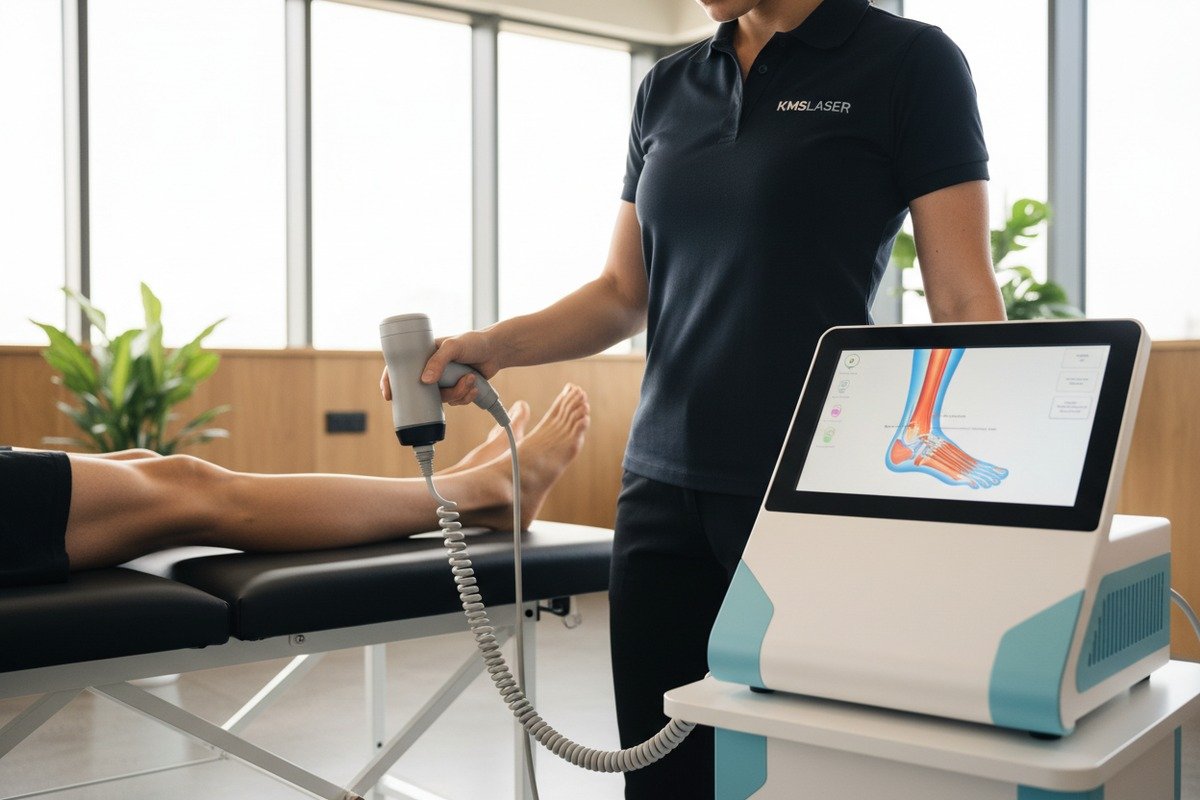
Every week, our production line ships shockwave therapy devices to clinics across three continents Extracorporeal shockwave therapy (ESWT) 1. Yet we still hear the same question from distributors: does this technology actually work for athletes? The frustration is real. You invest in equipment, your clients expect results, and mixed research findings create doubt.
Yes, shockwave therapy is effective for many sports injuries. Extracorporeal shockwave therapy (ESWT) delivers strong clinical evidence for conditions like plantar fasciitis, Achilles tendinopathy, and tennis elbow, with studies showing significant pain reduction and faster return to play compared to conservative treatments alone.
But effectiveness depends heavily on the device you use, the protocols you follow, and the quality of the equipment itself CE marking under MDR 2. Let me walk you through what matters most when sourcing shockwave therapy machines for your market.
How can I ensure the shockwave therapy machines I import deliver the clinical results my clients demand?
When we engineer our shockwave devices, the first question our R&D team asks is simple: will this actually help patients supplier qualification audits 3? You face the same challenge from the buyer's side. Importing machines that look impressive but fail to deliver clinical outcomes destroys your reputation fast.
To ensure imported shockwave machines deliver clinical results, prioritize devices with proven energy output consistency, validated treatment protocols, and proper certification. Request third-party test reports, demand treatment parameter documentation, and verify the manufacturer's quality management system before committing to orders.

Understanding What Drives Clinical Outcomes
The science behind shockwave therapy is straightforward. High-energy acoustic waves penetrate tissue and trigger biological responses. These include increased blood flow, neovascularization 4, collagen synthesis, and pain reduction. But here is the catch: these effects only happen when the device delivers the right energy at the right depth.
Our testing data shows that energy output variance above 15% between pulses leads to inconsistent treatment results. Cheaper machines often have variance rates of 25% or higher. This means one session might work well, while the next session underperforms. Your clients notice this inconsistency immediately.
Key Factors That Determine Treatment Success
| Factor | Why It Matters | What to Verify |
|---|---|---|
| Energy Output Stability | Consistent acoustic waves ensure predictable tissue response | Request pulse-by-pulse energy deviation reports |
| Penetration Depth Range | Different injuries require different depths (superficial vs. deep) | Confirm both radial and focused options available |
| Treatment Protocol Library | Pre-set protocols reduce operator error | Check if device includes evidence-based programs |
| Applicator Quality | Worn applicators lose energy efficiency | Ask about applicator lifespan and replacement costs |
Matching Device Capabilities to Injury Types
Not all shockwave devices treat all conditions equally. Plantar fasciitis 5 responds excellently to focused shockwave therapy, with studies showing pain scores dropping from 2.52 to nearly zero. But patellar tendinopathy shows more mixed results. This is partly about device capability and partly about protocol selection.
When our engineers calibrate machines for export, we configure treatment libraries based on the strongest evidence. For your market, you need devices that cover the most common sports injuries your clients treat. These typically include Achilles tendinopathy 6, tennis elbow, rotator cuff issues, and hamstring problems.
Certification and Clinical Validation
Regulatory approval does not guarantee clinical effectiveness, but it does indicate baseline safety and manufacturing standards. For the US market, FDA clearance 7 matters. For Europe, CE marking under MDR is essential. We hold both certifications, and our quality management system follows ISO 13485 standards 8.
Beyond certifications, ask potential suppliers for clinical study references. Reputable manufacturers can point you to published research using their specific devices. This separates serious manufacturers from those simply assembling components.
What technical specifications should I prioritize to guarantee my shockwave devices outperform cheaper competitors?
In our experience exporting to North American distributors, price objections come up constantly. Your customers compare your premium devices against budget imports from unknown factories. The difference is not visible on the outside. It lives in the technical specifications that determine long-term performance.
Prioritize maximum energy output (at least 5 bar for radial, 0.5 mJ/mm² for focused), frequency range flexibility, compressor durability rated for 10+ million pulses, applicator variety for different treatment depths, and intuitive software with customizable protocols. These specifications separate professional-grade equipment from disposable budget machines.

Energy Generation Systems Compared
The compressor is the heart of any radial shockwave device. Budget machines use compressors rated for 2-3 million pulses. Our standard production line uses compressors rated for 10 million pulses or more. This difference determines whether a device lasts two years or ten years.
For focused shockwave systems, the energy generation method matters even more. Electromagnetic generators 9 offer the best balance of precision and durability. Piezoelectric systems provide excellent accuracy but at higher cost. Electrohydraulic systems are older technology with more maintenance requirements.
Critical Specifications Comparison Table
| Specification | Budget Devices | Mid-Range Devices | Premium Devices |
|---|---|---|---|
| Max Pressure (Radial) | 2-3 bar | 4-5 bar | 5-6 bar |
| Compressor Life | 2-3 million pulses | 5-7 million pulses | 10+ million pulses |
| Frequency Range | 1-15 Hz | 1-20 Hz | 1-22 Hz |
| Applicator Options | 1-2 heads | 3-4 heads | 5+ heads |
| Protocol Memory | None or basic | Standard library | Customizable + updates |
| Warranty Period | 6-12 months | 12-18 months | 24+ months |
Software and User Interface Considerations
Our touchscreen interfaces display anatomical diagrams and treatment data in real-time. This is not just about looking modern. Clear visual feedback helps operators deliver consistent treatments. When your clients train new staff, intuitive software reduces errors significantly.
Look for devices that allow protocol customization. Evidence-based pre-sets are valuable, but experienced practitioners want to adjust parameters. The best systems offer both options. They also provide software updates as new research emerges.
Applicator Design and Versatility
Different injuries require different applicator sizes and shapes. A tennis elbow treatment uses a small, focused applicator. A hamstring injury needs a larger treatment area. Devices with limited applicator options force practitioners to compromise on treatment quality.
Our standard configuration includes five applicator heads covering treatment areas from 6mm to 35mm in diameter. This range handles everything from trigger points to large muscle groups. When you evaluate competitors, compare applicator variety and replacement costs.
Build Quality Indicators
Examine the housing materials. Premium devices use medical-grade plastics and metal components that withstand daily clinical use. Check the weight distribution. Well-engineered devices feel balanced and comfortable during extended treatments. Inspect cable connections and ports. Quality connectors last longer and maintain signal integrity.
How can I customize the design and branding of my shockwave equipment to fit my high-end market position?
When we work with brand owners in the US and Europe, they rarely want our standard appearance. Your market position depends on differentiation. A device that looks identical to ten other imports on Alibaba undermines your premium pricing. Our OEM and ODM services address this challenge directly.
Customize shockwave equipment through private labeling (your logo, colors, packaging), interface localization (language, measurement units, protocol names), and structural modifications (housing design, control layout). Work with manufacturers offering true ODM capabilities, not just sticker changes, to create genuinely differentiated products.
Levels of Customization Available
Customization ranges from simple cosmetic changes to complete device redesigns. Understanding these levels helps you match investment to market requirements.
| Customization Level | What It Includes | Typical MOQ | Lead Time |
|---|---|---|---|
| Basic Branding | Logo placement, color selection, packaging design | 10-20 units | 2-3 weeks |
| Interface Customization | Software language, measurement units, custom protocols | 20-50 units | 4-6 weeks |
| Housing Modifications | Panel colors, control knob styling, display bezels | 50-100 units | 8-12 weeks |
| Full ODM Development | Unique housing design, proprietary features, exclusive tooling | 200+ units | 16-24 weeks |
Branding Elements That Matter Most
The touchscreen interface is the primary interaction point. Custom boot screens, branded color schemes, and your company name throughout the software create immediate brand recognition. Our development team can implement these changes with minimal order quantities.
External branding includes logo placement on the main unit, applicators, and accessories. Consider the treatment handpiece especially. Practitioners hold this constantly. Your brand visible during every treatment reinforces identity with both operators and patients.
Packaging design often gets overlooked. A premium unboxing experience sets expectations before the device even powers on. Custom foam inserts, branded accessory pouches, and professional documentation reflect quality.
Color and Material Selection
Our standard production offers white main units with blue accent panels. But your brand colors matter. We can match Pantone colors for housing panels and produce custom-colored applicators. Material finish options include matte, gloss, and soft-touch textures.
Think about your target environment. Devices for high-end sports medicine clinics might use darker, more sophisticated color schemes. Equipment for general physiotherapy practices might benefit from brighter, more approachable appearances.
Proprietary Feature Development
True differentiation comes from exclusive features. Our engineering team can develop custom treatment protocols based on your clinical advisors' expertise. We can modify control interfaces to match your training programs. Some clients request proprietary applicator designs optimized for specific sports injury treatments.
These developments require investment but create genuine competitive advantages. Competitors cannot simply copy features that exist only in your product line. This protects your market position long-term.
Working Effectively With Manufacturers
Clear communication prevents costly mistakes. Provide detailed brand guidelines including exact color specifications, logo files in vector formats, and mockups of desired appearances. Request 3D renderings before production begins. Ask for physical samples of custom colors and materials.
Establish approval checkpoints throughout production. We provide photographs at key stages so you can verify branding accuracy before final assembly. This process catches errors early when corrections are simple and inexpensive.
What quality control steps will protect my brand from the risks of faulty shockwave therapy units?
Our quality control team catches defects that would devastate your reputation if they reached your customers. One malfunctioning unit at a prestigious sports clinic can undo years of brand building. The financial losses from returns, warranty claims, and lost accounts far exceed the cost of proper QC.
Protect your brand through supplier qualification audits, incoming component inspection, in-process monitoring at critical assembly stages, 100% functional testing before shipment, and pre-shipment inspection by independent third parties. Document everything and require corrective action reports for any defects found.

The True Cost of Quality Failures
When a shockwave device fails in the field, the direct costs include warranty replacement, shipping, and technician time. But indirect costs are larger. Your customer loses treatment revenue during downtime. Their patients experience treatment interruptions. Your sales team faces difficult conversations with prospects who heard about the failure.
We track warranty claim data carefully. Devices that skip testing steps have failure rates eight to ten times higher than fully tested units. The math is clear: comprehensive QC costs less than quality failures.
Critical Control Points in Production
Every shockwave device passes through specific checkpoints in our facility. Understanding these helps you ask the right questions when evaluating any supplier.
Pre-Production Quality Measures
Component inspection happens before assembly begins. We test compressors individually for pressure output and consistency. Circuit boards undergo automated optical inspection. Housings are checked for dimensional accuracy and surface defects.
Supplier qualification is ongoing, not one-time. We audit component suppliers annually and require certifications for critical parts. When a component fails incoming inspection, we quarantine the entire batch and investigate root causes.
In-Process Monitoring Checkpoints
| Assembly Stage | Tests Performed | Acceptance Criteria |
|---|---|---|
| Compressor Installation | Pressure output, cycle timing | Within 5% of specification |
| Electronics Assembly | Continuity, voltage regulation | Zero defects allowed |
| Software Loading | Boot sequence, protocol accuracy | Full functionality verified |
| Housing Assembly | Fit, alignment, seal integrity | Visual inspection pass |
| Final Assembly | Energy output calibration | Within 3% of target values |
Final Testing Protocols
Every unit undergoes a minimum 30-minute burn-in test simulating clinical use. We cycle through all pressure levels, frequency settings, and applicator configurations. This catches components that pass initial testing but fail under sustained operation.
Calibration verification uses reference instruments traceable to international standards. We record actual energy output values for each unit. This data ships with the device as proof of testing.
Third-Party Inspection Options
Many of our clients arrange independent pre-shipment inspections. Companies like SGS, Bureau Veritas, or Intertek send inspectors to our facility. They verify quantities, check specifications, and test random samples. This independent verification provides additional assurance before shipment.
We welcome these inspections and provide full access to testing equipment and documentation. Manufacturers who resist third-party inspection often have something to hide.
Documentation and Traceability
Every device has a unique serial number linked to complete production records. We track which components went into each unit, who performed each assembly step, and what test results were recorded. If a problem emerges months later, we can trace back to identify root causes and potentially affected units.
This traceability protects both parties. You can demonstrate due diligence to your customers. We can identify whether issues are isolated or systematic.
Conclusion
Shockwave therapy works for sports injuries when the equipment works properly. Your success depends on choosing devices with proven specifications, meaningful customization, and rigorous quality control. Partner with manufacturers who understand these requirements and can demonstrate their capabilities through documentation, testing data, and transparent processes.
Footnotes
1. Found a current and relevant article from Mayo Clinic on shockwave treatment for musculoskeletal care. ↩︎
2. Details the requirements and significance of CE marking for medical devices under EU MDR. ↩︎
3. Discusses the importance and process of supplier qualification audits in medical device manufacturing. ↩︎
4. Provides scientific evidence for shockwave therapy inducing neovascularization. ↩︎
5. Found a current Mayo Clinic article that lists plantar fasciitis as a condition treated by shockwave therapy. ↩︎
6. Discusses shockwave therapy for Achilles tendinopathy, including evidence and efficacy. ↩︎
7. Found the official FDA page explaining the Premarket Notification 510(k) process, which is the primary route for FDA medical device clearance. ↩︎
8. Explains ISO 13485 as the international standard for medical device quality management systems. ↩︎
9. Explains the physical principles and generation methods of electromagnetic shockwaves. ↩︎


